Isooctanoic Acid vs Isononanoic Acid vs
2-Ethylhexanoic Acid: Key Differences & How to Choose
Structure · Properties · Regulatory status · Application performance · Substitution guide · Procurement checklist
🔗 View Isooctanoic Acid Product Page📋 Table of Contents
- The Naming Confusion: What Each Term Actually Means
- Structural Comparison at a Glance
- Physical & Chemical Property Comparison
- Regulatory Comparison: The Critical H361 Difference
- Application Comparison: Coating Driers
- Application Comparison: PVC Stabilisers
- Application Comparison: Lubricants & MWF
- Substitution Guide: When & How to Switch
- Procurement Decision Checklist
- Frequently Asked Questions
🏷️ 1. The Naming Confusion: What Each Term Actually Means
Before comparing the three acids, it is essential to establish precise definitions - because the naming of these compounds in commercial and regulatory contexts is genuinely confusing, and ordering the wrong product because of a naming ambiguity is a real and recurring problem in industrial procurement.
| CAS (mixture) | 25637-84-7 |
| Definition | Commercial mixture of branched C8 fatty acid isomers |
| Main component | 2-EHA (85–95%) |
| Formula | C₈H₁₆O₂ |
| MW | 144.21 g/mol |
The trade/commercial name. "Isooctanoic acid" refers to a mixture - the isomeric composition varies by producer and process.
| CAS | 149-57-5 |
| Definition | Single pure isomer: 2-ethyl substituted hexanoic acid |
| IUPAC name | 2-Ethylhexanoic acid |
| Formula | C₈H₁₆O₂ |
| MW | 144.21 g/mol |
The IUPAC name for the specific dominant isomer in IOA. "2-EHA" and "isooctanoic acid" are used interchangeably in practice - for most industrial applications they are the same product.
| CAS | 26896-18-4 |
| Definition | Different product: commercial mixture of branched C9 fatty acid isomers |
| Major isomer | 3,5,5-Trimethylhexanoic acid (~60–75%) |
| Formula | C₉H₁₈O₂ |
| MW | 158.24 g/mol |
A genuinely different product - one carbon longer, different branching pattern. Not synonymous with IOA or 2-EHA. The "iso-" prefix in both names causes confusion but they are distinct compounds.
⚠️ The most common procurement error: Ordering "isooctanoic acid" when "isononanoic acid" (or vice versa) is required - or ordering "2-ethylhexanoic acid" thinking it is a different product from "isooctanoic acid." Always specify both the product name and the CAS number in every purchase order. If a supplier's product has a CAS number that does not match what you are ordering, request clarification before proceeding.
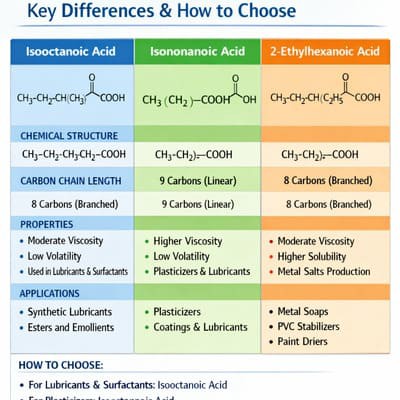
🧬 2. Structural Comparison at a Glance
|
CH–CH₂–CH₃
|
COOH
|
CH₂–COOH
[3,5,5-trimethylhexanoic]
🔑 The One Structural Difference That Drives Everything Else
The single most important structural distinction is where the branch sits relative to the carboxylic acid group. In 2-EHA/IOA, the ethyl branch is directly adjacent to the –COOH (α-position, C2). In the major isomer of INA (3,5,5-trimethylhexanoic acid), the nearest branch is at the β-position (C3), with a further neo-type grouping at the far end of the chain. This α-vs-β branching difference explains why: (1) 2-EHA/IOA provides better steric protection of the metal–oxygen bond in derived metal soaps → better hydrolytic stability; (2) INA-based metal soaps have slightly higher bulk hydrophobicity (longer chain) → marginally better oil solubility in very non-polar media; and (3) 2-EHA/IOA carries the H361 reproductive toxicity classification while INA does not.
📊 3. Physical & Chemical Property Comparison
| Property | IOA / 2-EHA (C8) | Isononanoic Acid (C9) | Practical Consequence |
|---|---|---|---|
| Molecular weight | 144.21 g/mol | 158.24 g/mol | IOA has 9.7% lower MW; delivers more moles of carboxylate per gram → higher metal loading per gram acid in metal soap synthesis |
| Acid value (mg KOH/g) | ~385 | ~354 | AV difference of ~8.4%; need ~8.4% more INA by mass to react with the same metal oxide charge → always recalculate batch stoichiometry when switching |
| Melting point | −59 °C | ~−50 to −55 °C | Both fully liquid at all practical temperatures; no handling difference |
| Boiling point | 228 °C | ~250–260 °C | INA slightly higher BP → marginally lower vapour pressure; both have negligible VP at room temperature |
| Density (20 °C) | 0.902–0.910 g/cm³ | 0.898–0.905 g/cm³ | Very similar; INA marginally less dense; negligible practical difference for volume-based dosing |
| Refractive index (nD²⁰) | 1.424–1.430 | 1.432–1.438 | ⭐ Key distinguishing test: RI difference of ~0.007–0.008 clearly separates IOA from INA on an Abbe refractometer |
| pKa (aqueous) | ~4.85 | ~4.90 | Negligible difference; both are weak carboxylic acids with very similar metal salt formation kinetics |
| log P (octanol/water) | ~3.05 | ~3.50 | INA is more hydrophobic; INA-based metal soaps have slightly better solubility in very non-polar base oils; IOA-based soaps are slightly better in mildly polar solvents |
| Branching position | α-Ethyl (C2) | β-Methyl (C3) + remote neo | IOA's α-branch gives better steric protection of metal–O bond → better hydrolytic stability of Co/Mn/Zr isooctanoates vs INA analogues |
| Water content of metal soaps (hydrolytic stability) | Good (α-branch shield) | Good to slightly better (longer chain) | Practical difference is small in most applications; both adequate for drier and stabiliser use |
| Metal content of derived salt (per gram acid) | Higher ⭐ (C8 lower MW) | ~8% lower per gram | To prepare Co drier at 6% Co: need 8% less IOA by mass than INA for same molar Co loading |
⚠️ 4. Regulatory Comparison: The Critical H361 Difference
The most practically significant difference between IOA/2-EHA and isononanoic acid is not chemical or physical - it is regulatory. 2-Ethylhexanoic acid (and by association, commercial isooctanoic acid containing ≥85% 2-EHA) carries a reproductive toxicity classification (H361) under EU CLP. Isononanoic acid does not. This single regulatory distinction is increasingly driving substitution decisions in the European coatings, plastics, and lubricant industries.
| Regulatory Criterion | IOA / 2-EHA | Isononanoic Acid |
|---|---|---|
| EU CLP Reproductive Toxicity | Repr. Category 2, H361 ⚠️ Suspected of damaging fertility or the unborn child |
Not classified ✅ |
| EU REACH SVHC Status | 2-EHA (CAS 149-57-5) on SVHC Candidate List ⚠️ | Not on SVHC Candidate List ✅ |
| REACH Annex XIV (Authorisation) | Possible future restriction / authorisation process (SVHC pathway) | No restriction anticipated ✅ |
| Downstream Article 33 Obligation | If >0.1% w/w in article → notify customers (SVHC) ⚠️ | No Article 33 obligation ✅ |
| SDS GHS Section 2 Signal Word | WARNING + H361 hazard statement ⚠️ | WARNING (skin/eye irritant only) ✅ - cleaner SDS |
| Workplace Risk Assessment | Specific reproductive hazard risk assessment required; women of childbearing potential must be specifically assessed ⚠️ | Standard COSHH/REACH risk assessment; no specific reproductive concern ✅ |
| Consumer/Retail Product Use | May be restricted/prohibited in some consumer applications under EU REACH Annex XVII ⚠️ | No REACH Annex XVII restriction ✅ |
| US TSCA Status | Active inventory; no Section 6 action currently; no SNUR | Active inventory; no action; slightly cleaner regulatory profile |
⚠️ The H361 issue in practical terms: For EU-based formulators and manufacturers, the H361 classification on IOA/2-EHA creates growing compliance burdens: SDS Section 2 must prominently declare H361; workplace risk assessments must specifically address reproductive hazards; women of childbearing potential working with IOA require dedicated risk assessment and potentially restricted access or enhanced controls; downstream customers (particularly in consumer goods, toys, food contact, and cosmetic-adjacent industries) may require a declaration or substitution. For businesses that can achieve equivalent technical performance with INA, the regulatory argument for substitution is compelling and growing stronger as REACH enforcement tightens.
🎨 5. Application Comparison: Coating Driers
Coating drier synthesis is the largest commercial application for both IOA and INA. Cobalt, manganese, zirconium, and calcium isooctanoates and isononanoates are commercially produced driers, and the two acid families are directly competitive in this market. The choice between them is driven by performance requirements, regulatory compliance, cost, and formulation tradition.
| Drier Parameter | IOA-Based Drier (Co/Mn/Zr isooctanoate) | INA-Based Drier (Co/Mn/Zr isononanoate) |
|---|---|---|
| Surface dry time (alkyd, 0.04% Co) | Reference (e.g. 2.5 h) | Comparable (±10–15% at equal Co mass; same at equal Co moles) |
| Through dry time | Reference | Comparable; slightly better in high-humidity conditions (more hydrophobic environment) |
| Hydrolytic stability of drier solution | Good (α-ethyl protection) | Good to slightly better (longer chain bulk) |
| Metal content per gram acid (Co example) | Higher ⭐ (C8) | ~8% lower Co per gram acid (C9, higher MW) |
| Stoichiometry adjustment when switching | Reference | Use 8–10% more INA by weight to achieve same molar metal loading |
| Regulatory preference (EU) | ⚠️ H361 on 2-EHA component; SVHC pressure increasing | ✅ No H361; preferred for EU compliance-driven reformulation |
| Industry tradition | ⭐ Dominant established standard; most drier specifications reference isooctanoate | Growing but established in some markets (ExxonMobil Exxal 9 based); requires customer approval for specification change |
| Cost (typical) | Reference ⭐ (often slightly lower) | Comparable to slightly higher (depends on regional market) |
🧱 6. Application Comparison: PVC Stabilisers
Both Ca/Zn isooctanoate and Ca/Zn isononanoate systems are commercially used in PVC heat stabilisers. The choice is analogous to the drier case - the chemistry is equivalent, but the stoichiometry and regulatory profile differ.
- HCl scavenging capacity per gram of Zn soap: Zn isooctanoate has ~8% higher HCl capacity per gram than Zn isononanoate (higher AV → more Zn per gram). Use 8% more isononanoate by mass to achieve equivalent Zn loading in the stabiliser system.
- Colour of stabiliser solution: Both give colourless to very pale yellow Ca/Zn solutions - no practical difference for PVC appearance.
- Compatibility with plasticisers: Both are compatible with DOTP, DINCH, and ESBO in liquid one-pack formulations.
- Long-term thermal stability of PVC: Essentially equivalent when used at equimolar metal loading.
- Regulatory preference: INA-based Ca/Zn stabilisers are increasingly preferred for EU consumer-contact PVC (cables, toys, food packaging film) where H361-free supply chain is required.
Zn(IOA)₂: Zn content = 21.5% (theoretical)
Zn(INA)₂: Zn content = 20.6% (theoretical, lower MW/Zn)
→ Use 4.4% more Zn(INA)₂ by weight for same Zn loading
For calcium component:
Ca(IOA)₂: Ca content = 13.8% (theoretical)
Ca(INA)₂: Ca content = 12.7% (theoretical)
→ Use 8.7% more Ca(INA)₂ by weight for same Ca loading
Adjust the one-pack formula to maintain the same phr Ca and Zn levels in the final PVC compound. The change is purely in the mass of the stabiliser components - the PVC processing behaviour and end properties are unchanged when metal loadings are equivalent.
🔧 7. Application Comparison: Lubricants & MWF
| Lubricant Application | IOA Performance | INA Performance |
|---|---|---|
| Corrosion inhibition (neat oil) | Good - adsorbs on ferrous surfaces effectively | Good to slightly better - longer chain gives denser adsorbed film ⭐ |
| Corrosion inhibition (water-based MWF) | Good (as amine salt); higher carboxylate density | Good (as amine salt); slightly more hydrophobic barrier |
| Mo/Sb metal soap EP additives | Established standard ⭐; Mo isooctanoate widely commercially available | Mo isononanoate also available; slightly more hydrophobic; similar EP performance |
| Oil solubility in very non-polar base oil (PAO, mineral) | Excellent | Excellent; marginally better at very low temperatures due to more hydrophobic C9 chain ⭐ |
| Regulatory preference (EU lubricant) | ⚠️ H361 on IOA; requires workplace reproductive risk assessment | ✅ No H361; growing preference in EU lubricant formulations |
| Bioresistance in MWF | Standard; susceptible to biodegradation in aqueous MWF without biocide | Similar; slightly more hydrophobic chain may offer marginally better bioresistance but both require biocide in MWF |
🔄 8. Substitution Guide: When & How to Switch
- EU regulatory compliance: eliminate H361 from your SDS and reduce REACH SVHC obligations
- Customer supply chain requirements: customer demands H361-free formulation for consumer goods
- Women of childbearing potential work with the material: eliminate reproductive hazard risk assessment obligation
- Application is regulatory-sensitive: toys, food contact, medical devices, cosmetic packaging
- REACH authorisation uncertainty: hedge against potential future IOA/2-EHA restrictions
- Customer specification explicitly requires "isooctanoate" or CAS 25637-84-7 → need formal specification change approval
- Proprietary drier or stabiliser formula registered with regulatory body under IOA/2-EHA → requires re-registration
- Application requires exact acid value match → INA AV is ~8% lower; recipe must be recalculated
- Cost difference is significant and technical equivalence is not required by customer
- Obtain COA for INA batch: note actual acid value (typically 348–360 mg KOH/g)
- Recalculate metal oxide charge: Co(OH)₂ charge = (mass of INA ÷ EW of INA) × (MW of Co(OH)₂ ÷ 2)
- Adjust total acid charge: mass of INA = mass of IOA × (AV of IOA ÷ AV of INA)
- Confirm target metal% in product by ICP after synthesis trial batch
- Run application performance trial: drier (dry time panel test); stabiliser (Congo Red test); lubricant (corrosion + wear test)
- Update SDS: remove H361; update CAS number; confirm EC number
- Notify customers if the final product specification changes (metal%, performance data)
📋 9. Procurement Decision Checklist
| Procurement Question | Answer → IOA | Answer → INA | Decision Logic |
|---|---|---|---|
| Is your customer or product standard specified as "isooctanoate" / CAS 25637-84-7? | ✅ Yes | ❌ No | Follow specification; get approval before changing acid type |
| Is H361 reproductive toxicity a concern in your workplace or supply chain? | ❌ Yes → burden | ✅ No → cleaner | Favour INA if H361 is a regulatory or occupational health concern |
| Is maximising metal loading per kg of acid important (cost efficiency in drier/stabiliser synthesis)? | ✅ IOA wins (~8% more metal/kg) | ❌ Less efficient | IOA delivers more moles of carboxylate per kg; lower metal oxide consumption per kg acid |
| Is your application in EU consumer goods, toys, or food-contact materials? | ⚠️ SVHC concern | ✅ Safer choice | Consumer regulations increasingly restrict SVHC; INA avoids this issue |
| Is technical performance in hydrolytically demanding conditions (high humidity, outdoor exposure) a key requirement? | ✅ Slight edge (α-branch) | Comparable | Marginal advantage for IOA-based metal soaps; confirm with formulation testing |
| Is the application in a non-EU market (Asia, Middle East, Americas) with no H361 pressure? | ✅ Cost/tradition advantage | No regulatory advantage | Outside EU, H361 is less of a driver; IOA dominates as the lower-cost established standard |
| Does your incoming QC need to reliably distinguish IOA from INA? | Use refractive index (IOA: 1.424–1.430 vs INA: 1.432–1.438) AND acid value (IOA: ~385 vs INA: ~354) - two tests clearly separate the two products | Both tests together provide unambiguous identification; RI alone is marginal; AV alone is marginal; combined = definitive | |
📚 Related Articles
❓ 10. Frequently Asked Questions
Q1: Is isononanoic acid the same as isooctanoic acid?
No - isononanoic acid (INA, CAS 26896-18-4) and isooctanoic acid (IOA, CAS 25637-84-7) are different products. They are both branched-chain carboxylic acids from the same "iso-acid" family produced by the Koch/oxo reaction, and they are used in overlapping applications, but they have different chain lengths (C9 vs C8), different molecular weights (158.24 vs 144.21 g/mol), different acid values (~354 vs ~385 mg KOH/g), and critically different regulatory profiles (INA has no H361 reproductive toxicity classification; IOA does through its dominant 2-EHA component). The confusion arises because both use the "iso-" prefix and are sometimes loosely grouped as "iso-acids" in trade discussions. Always specify the CAS number to eliminate ambiguity: 26896-18-4 for isononanoic acid; 25637-84-7 for isooctanoic acid / 149-57-5 for 2-ethylhexanoic acid.
Q2: What is the market for isononanoic acid and who produces it?
Isononanoic acid is a commercially significant specialty fatty acid produced primarily by the Koch reaction (carbonylation of C8 olefins from isooctene/disobutylene feedstock). Major producers include BASF (under the Versatic / Lutensol / BASF chemical portfolio), ExxonMobil (Exxal 9 acids), and several Chinese producers who have expanded INA capacity significantly. The global INA market serves coating driers, metallic soap lubricant additives, polyester and alkyd synthesis, and PVC stabiliser applications. INA has gained market share over IOA in the EU market particularly as the H361 regulatory pressure on IOA/2-EHA has increased - this is the primary growth driver for INA as a technically equivalent but regulatorily cleaner alternative. Market search volume for "isononanoic acid" (590 searches/month) exceeds that for "isooctanoic acid" (20 searches/month) in global SEO data, reflecting the growing commercial interest in INA as a procurement category.
Q3: How do I test whether I have received isooctanoic acid or isononanoic acid?
The most reliable two-test incoming QC protocol to distinguish IOA from INA: (1) Acid value titration (ASTM D974): IOA = 375–395 mg KOH/g; INA = 342–365 mg KOH/g. A clear numerical difference (~8–10%) separates the two. Test takes 20–30 minutes with standard titration equipment. (2) Refractive index (Abbe refractometer, 20 °C): IOA = 1.424–1.430; INA = 1.432–1.438. The ~0.007 difference is clearly measurable. Test takes 5 minutes. Together these two measurements provide confident, definitive identification without expensive instrumentation. If both values match the expected range for your specified product → accept. If either value is out of range → reject and request explanation from supplier. GC-MS provides the gold standard confirmation of identity and isomer composition but is slower and requires specialist equipment.
Q4: Why does isononanoic acid not have the H361 classification that isooctanoic acid has?
The H361 reproductive toxicity (Category 2, suspected) classification applies specifically to 2-ethylhexanoic acid (CAS 149-57-5) based on animal studies demonstrating effects on reproductive organs and development at relevant exposure doses. The key structural feature that appears to drive this toxicity is the α-ethyl branch at C2 of the carboxylic acid - the specific geometry of 2-EHA relative to metabolic activation pathways in mammals. Isononanoic acid (predominantly 3,5,5-trimethylhexanoic acid, with a β-methyl branch and remote neo grouping) has a different branching pattern, a longer chain, and apparently does not share the metabolic pathway that leads to reproductive toxicity observed with 2-EHA. The regulatory studies supporting the H361 classification were conducted on the racemate of 2-EHA; isononanoic acid has not received an equivalent H361 classification in EU CLP studies. This is not simply because INA has been less studied - it has been specifically assessed and not found to trigger equivalent classification. The structural biology underlying this difference remains an active area of research, but the practical regulatory outcome is clear: INA = no H361; IOA/2-EHA = H361.
Q5: What are the isononanoic acid uses and how do they compare to isooctanoic acid?
Isononanoic acid's industrial uses closely mirror those of isooctanoic acid because the two are structurally analogous and functionally similar branched fatty acids. The principal uses of INA are: (1) Metal soaps for coating driers - Co, Mn, Zr, Ca isononanoates are commercial drier products; (2) PVC stabiliser components - Ca/Zn isononanoate in liquid one-pack stabilisers; (3) Lubricant additives - Mo, Zn isononanoates as EP and corrosion inhibitor precursors; (4) Polyester and alkyd synthesis - INA contributes branched-chain flexibility to polyester backbones; (5) Metallic soap thickeners - aluminium and lithium isononanoates in specialty greases. The primary advantage of INA over IOA in all these applications is regulatory (no H361); the primary advantage of IOA over INA is acid value efficiency (higher moles of carboxylate per gram, lower stoichiometric metal oxide requirement). In non-EU markets where H361 is not a driver, IOA and INA compete primarily on price and established supply relationships.
Q6: Can Sinolook supply both isooctanoic acid and isononanoic acid?
Yes - Sinolook Chemical supplies both isooctanoic acid (CAS 25637-84-7) and isononanoic acid (CAS 26896-18-4) for industrial applications. For customers considering switching from IOA to INA for regulatory compliance reasons, we can provide samples of both products simultaneously for direct formulation comparison, along with full COA documentation (acid value, APHA colour, water content, refractive index per batch) and GHS SDS in your language. For EU buyers, we provide REACH OR letters for both products. Contact us via WhatsApp (0086 18150362095), WeChat/Tel (0086 13400715622), or email (sales@sinolookchem.com) to discuss your specific requirements and receive quotations for both products.
Source IOA or Isononanoic Acid - Compare Both
Contact Sinolook Chemical
Isooctanoic acid (CAS 25637-84-7) · Isononanoic acid (CAS 26896-18-4)
Full COA per batch · REACH OR for EU buyers · Side-by-side samples available · Export to 50+ countries